Summary
Health Innovation Oxford and Thames Valley (HIOTV), in partnership with Treatment Technologies & Insights (TTI) and Bayer Plc, undertook a collaborative project from August 2023 to June 2025 to assess the feasibility, implementation readiness and adoption requirements for Wave Health Pro – a digital health platform customised to support men living with prostate cancer. Wave Health is a proactive patient risk management software (app and portal) that operationalises patient-reported data to optimise care coordination. The platform is owned by TTI. Bayer Plc partially funded prostate cancer-specific development work and supports marketing of the disease-agnostic product in the UK on TTI’s behalf. Wave Health is a standalone product, not connected with any medicinal products.
The platform enables patients to track symptoms, medications, laboratory results, side-effects and lifestyle information with personalised guidance via a mobile application. Clinicians can remotely monitor patient-reported outcomes through a connected care portal to support communication, clinical decision-making and identification of potential deterioration. The shared patient-clinician interface supports earlier intervention and proactive risk stratification, while enabling patients to play a more active role in managing their condition.
The work highlighted key preconditions for digital health solutions to succeed in prostate cancer care, including UK-specific clinical and economic evidence, regulatory readiness, NHS IT system integration and clear funding models.
Declaration of interests and roles:
Wave Health is manufactured by Treatment Technologies & Insights (TTI). Health Innovation Oxford & Thames Valley (HIOTV) was sponsored by Bayer Plc to conduct an independent feasibility study on the TTI technology. Bayer Plc partners with TTI solely as the UK marketing agent for the prostate cancer version of Wave Health and is not responsible for the product’s content or functionality.
Why this matters
Prostate cancer is the most common cancer in men, with over 52,000 new diagnoses annually in the UK[1]. As survival improves, more men need personalised, long-term support amid increasing service pressures.
Wave Health Pro offers a scalable and customisable digital solution which is purpose-built for prostate cancer but adaptable across conditions, promoting self-management, enhancing communication between patients and clinicians and enabling remote monitoring. This aligns with the NHS Long Term Plan’s vision for digitally enabled, data-driven care[2]. By improving communication and automating routine tasks, the platform facilitates care coordination, reduces clinician workload and optimises resource use, driving measurable gains in efficiency, patient engagement and overall quality of care. Bayer’s collaboration with Wave Health reflects its commitment to innovations that enhance patient experience and outcomes, underpinned by real-world evidence and sustainable adoption[3].
Although this project concluded prior to the publication of the NHS Medium-Term Planning Framework (2026/27-2028/29)[4], its findings remain highly relevant, as the Framework now reinforces the importance of digital monitoring and electronic PROMs as core enablers of productivity, patient-centred care and digital transformation across the NHS.
[1] https://prostatecanceruk.org/get-involved/campaigning/our-manifesto-for-the-next-government#:~:text=harder%20to%20treat.-,The%20problem,health%20care%20inequalities%20even%20worse
[2] https://www.gov.uk/government/publications/10-year-health-plan-for-england-fit-for-the-future/fit-for-the-future-10-year-health-plan-for-england-executive-summary
[3]https://www.mckinsey.com/~/media/mckinsey/dotcom/client_service/pharma%20and%20medical%20products/pmp%20new/pdfs/beyond_the_pill_creating_medical_value_through_technology_enablement.ashx?utm_source=chatgpt.com
[4] NHS Medium-Term Planning Framework (2026/27-2028/29)
 What we did
What we did
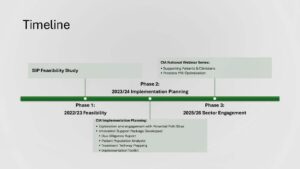
The project was delivered in three phases which aimed to assess stakeholder needs and feasibility of the technology, plan for implementation and engage with the sector to explore the potential of digital solutions in prostate cancer care. The table (right) outlines the project delivery timeline.
Phase 1: Feasibility study
Using the Lean Assessment Process (LAP) methodology, the HIOTV Strategic and Industry Partnerships (SIP) team interviewed 15 clinical and commissioning stakeholders across seven NHS trusts. Analysis of responses and human-factors mapping highlighted a strong unmet need for digital tools in prostate cancer follow-up. Stakeholders viewed Wave Health Pro as a potential advancement in efficiency and patient-centred care but emphasised the need for UK-specific real-world and economic evidence to demonstrate impact.
Read more about the feasibility study
Phase 2: Implementation planning
Building on the findings from Phase 1, Phase 2 aimed to progress from feasibility to implementation readiness. Using an engagement-based approach, the HIOTV Clinical Innovation Adoption (CIA) team held discussions with several NHS trusts to identify opportunities to pilot the technology and assess its potential impact within prostate cancer care. These discussions confirmed strong clinical interest and recognition of the platform’s potential to enhance patient follow-up and service efficiency. However, uncertainty around sustainable funding beyond pilot phases was also identified as a key barrier to adoption. Trusts expressed willingness to adopt the platform if long-term funding were to be available, highlighting the need for a clear, sustainable commercial model extending beyond short-term pilots and demonstrating measurable value for the wider healthcare system. The team noted that TTI was working through regulatory requirements while some aspects required additional work to facilitate access to the NHS market.
In response, the project pivoted to focus on identifying key enablers for NHS adoption and clarifying what would be required for the platform to achieve NHS market readiness. This shift generated valuable learning which was translated into a tailored innovation support package for the industry partner, comprised of a Due Diligence Report, Population Health Analysis, a Treatment Pathways Review and an Implementation Toolkit. These outputs addressed regulatory, clinical, population and implementation considerations for Wave Health Pro and were developed through desk-based research, drawing on published evidence, policy documents, national datasets and NHS digital health frameworks.
Phase 2 gave Bayer Plc and TTI a clear understanding of NHS adoption requirements and offered a clear roadmap to implementation as the Innovation Support Package outlined practical steps required to progress towards adoption readiness.
Phase 3: Sector engagement
To sustain momentum and build national awareness, HIOTV hosted a webinar series on innovation in prostate cancer care. The sessions brought together clinicians, service managers and innovation leads from across the UK to discuss service challenges and explore how digital and patient-centred solutions can improve pathways and outcomes.
The webinars showcased examples of innovation in diagnosis, treatment and follow-up care, fostering collaboration and shared learning across sectors. By facilitating national dialogue, this phase contributed to the national conversation on digital transformation in prostate cancer care.
Key findings and recommendations
The work identified clear opportunities alongside systemic barriers.
Wave Health Pro aligns with NHS priorities for personalised, digital-first care. Platforms of this kind can empower patients, enable remote monitoring and free up clinical capacity. Population analysis suggests significant potential for scale, positioning prostate cancer care as a suitable pathway for early adoption of digital health solutions.
Stakeholders were positive but cautious. NHS organisations require robust UK-specific evidence, including health-economic data, before committing to sustained use. Funding uncertainty remained the central concern: many Trusts asked, “If we try it, can we keep it?”, and integration with IT systems, workforce readiness, workflow fit and regulatory clarity (including UKCA marking) as key adoption requirements prior to piloting.
To progress from interest to adoption, it was recommended Wave Health Pro:
- look to generate real-world UK evidence and economic data to demonstrate value
- develop flexible pricing models aligned with NHS budgets
- ensure seamless technical and workflow integration within clinical pathways
- clarify its regulatory status and achieve data governance compliance.
Establishing clear market positioning that highlights the platform’s unique benefits further strengthens its adoption case across the NHS.
Feedback
“This collaboration has been instrumental in strengthening how Wave Health aligns with NHS priorities, particularly around digitally enabled, data-driven care and proactive patient management. The insights gained have helped ensure the platform supports scalable, sustainable adoption while delivering meaningful value for both patients and healthcare systems.” – Wave Health CEO & founder Matt Lashey
Conclusion
The project assessed the feasibility, implementation requirements and adoption challenges for Wave Health Pro within NHS prostate cancer pathways. It confirmed the need for UK-specific evidence, regulatory clarity and sustainable funding before large-scale deployment can occur. The findings provide a practical understanding of the steps required for Wave Health Pro and similar digital health solutions to move from concept to implementation across the NHS.
Post project updates and external publications
This section contains developer-reported updates provided after completion of the HIOTV project. These were not evaluated or independently verified by HIOTV and are included for context only.
Regulatory and compliance update
Since project completion, Wave Health Pro has successfully addressed previously identified regulatory and clinical compliance requirements – removing a major barrier to adoption. The platform is now an MHRA-registered Class-1 Software as a Medical Device (SaMD) in the UK, designed to enable proactive risk management among patient populations.
The platform fully complies with NHS Digital Technology Assessment Criteria (DTAC) and maintains Cyber Essentials certification. It is in full compliance with DCB0129 clinical safety and risk management standards. It operates under a Data Protection Impact Assessment (DPIA), hazard log, and Clinical Safety Case Evaluation/Reports – meeting NHS patient safety and governance requirements. Additionally, Wave Health’s ISO 13485-compliant Quality Management System (QMS) and ISO 27001-compliant Information Security Management System (ISMS) are currently undergoing notified body certification.
Integration update
Since project completion, Wave Health has introduced a flexible integration model that allows trusts to choose the level of connectivity and workflow alignment that matches clinical goals and technical capacity. Options range from lightweight single sign-on (SSO) to one-way data sharing, to advanced bi-directional interoperability. The Wave Health Care Portal is a web application and can be embedded in any system that supports a web view or launched standalone with SSO.
External publications
In addition to the HIOTV-led project, an independent publication concerning Wave Health Pro has appeared in the ESMO Journal, Real World Data and Digital Oncology: https://www.esmorwd.org/article/S2949-8201(2500066-9/fulltext
This publication is not affiliated with HIOTV and offers peer-reviewed analysis of the platform’s feasibility and role in digital oncology through a study conducted in eight Spanish hospitals.
Additional resource
Further information on Wave Health Pro and its UK offering is available via Wave Health UK’s website: https://www.wavehealth.app/wave-uk
Acknowledgements
We gratefully acknowledge Bayer Plc and TTI for their partnership and flexibility and thank the clinicians and contributors who shared their insights and experience throughout the project.
Contact: Marianna Lepetyukh, Senior Programme Manager. Marianna.Lepetyukh@healthinnovationoxford.org
